 A materials management system plays a vital part of any hospital because it is accountable for all the inventory that is stored inhouse and is the source of information pertaining to any material that is used in a hospital. These systems, like PeopleSoft, Lawson or Infor, manage vendor contracts, maintain pricing, identify consigned or owned supplies, release purchase orders, and manage par levels. However, it currently requires considerable manual effort to count inventory on hand, compare it with set Par levels, and enter the information for processing. This prevents the complete automation of the inventory management process. Hospital staff waste valuable time doing these redundant tasks which could be better spent doing critical hospital work. This also makes the system prone to manual-entry errors.
A materials management system plays a vital part of any hospital because it is accountable for all the inventory that is stored inhouse and is the source of information pertaining to any material that is used in a hospital. These systems, like PeopleSoft, Lawson or Infor, manage vendor contracts, maintain pricing, identify consigned or owned supplies, release purchase orders, and manage par levels. However, it currently requires considerable manual effort to count inventory on hand, compare it with set Par levels, and enter the information for processing. This prevents the complete automation of the inventory management process. Hospital staff waste valuable time doing these redundant tasks which could be better spent doing critical hospital work. This also makes the system prone to manual-entry errors.
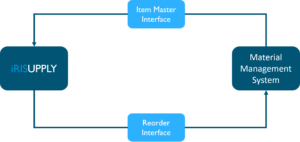
Many hospitals utilize a point of use solution to track implant and supply usage in the procedural areas, like the Operating Rooms, Cath Lab or Interventional Radiology. These systems, like iRISupply, can seamlessly integrate with the hospital’s materials management system. Successful integration can lead to automated workflows with minimum human intervention, reducing the amount of work required and eliminating errors from manual entry.
The process of integration development, also known as interface development, between two systems is a collaborative effort. It requires commitment and cooperation between owners of the two systems to do their part as planned in a timely manner. An integration project can drag on and eventually fail if one of the stakeholders does not commit dedicated resources to the project. The process of integration consists of four main phases:
- Kick-off meeting / Planning
- Design and Development
- Testing
- Deployment on Production / Go Live
The initial phase of integration development begins with the Kick-off meeting where the interface teams from both parties meet to discuss project requirements and plan the project schedule. Both teams exchange the data specification, which is integral to the process because it lays out the foundation of what is to be shared within both systems.
Based on the data specification, both teams begin the Design and Development phase. Many times, we use the preferred method of common interface specifications (e.g. HL7, RQ500, etc.) because this will speed up the design and development process. Custom specification can also be used when developing custom/uncommon interface to meet specific requirements. This, however, will take more time to design and develop as both parties need to agree on the data specification, format and communication protocol. In this phase, samples of interface messages are exchanged and used to verify and validate the interface scripts that are being developed. Upon completion of verification and validation, the interface scripts are released for site testing.
In the Testing phase, the interface scripts will be tested against different scenarios and events that may occur during real operation. These scenarios and events are defined in the test cases that are described in detail following the test plan, which usually comes from the hospital interface team. The duration of the Test will depend on the type of interface, the complexity of the use cases, the number of test iterations required and resource availabilities. Typically, the testing sessions will include: connectivity test, basic test cases, exception test cases and 3 iterations/revisions.
After Testing is completed, Deployment on Production is planned by considering: system down time during deployment (this may impact daily operations), user training and adjustments/bug fixes during transition. Both teams will monitor the interface closely for a period of time until the interface is performing as designed and is stable, after which the interface is declared to be Live. At this point the project is completed and the maintenance work is handed over to a support team.
Integration with the materials management system usually involves development of the following interfaces:
- Item Master Update Interface: this interface automates synchronization of product data information from the materials management system (the source of truth) with other systems. This ensures that information used in daily operations is always consistent and up to date, thus removing redundant data entry by users that can be prone to errors and inaccuracy. Key information that is typically managed/maintained in materials management systems include: Product Cat Number, Description, Manufacturer, Unit Price, Package Quantity, Order Unit of Measure, Charge Code, Par Levels, etc.
- Reorder Interface: this interface performs order calculations based on inventory data, Par levels (min/max) and usage characteristics to determine the precise list of items to be ordered. The purpose of this interface is to ensure that there is a perfect balance of inventory in stock – not too few, which can result in stock outs, and not too many, which can lead to unnecessary waste. Advanced inventory systems like iRISupply from Mobile Aspects use RFID technology to automate inventory count calculations and capture usage characteristics automatically, removing the human element from this process. User involvement is still needed to approve the order from the recommended list to provide control and accountability of the process. Upon approvals, order requisition messages will be automatically generated and sent out to the Materials management system for Purchase Order generation.
In my experience, maintaining a materials management interface is relatively easy. Aside from the occasional upgrade or customization request from a customer, a materials management system does not often change frequently.
Integration with a materials management system, in particular the Item Master Update Interface, is very essential, as it provides the groundwork and pre-requisite for all other interfaces. Once we have an Item Master Update Interface, not only can we can build other materials management interfaces, such as the Reorder Interface, but it is also required to build interfaces with other systems in the hospital, such as billing and usage interfaces.
The Item Master Interface and a strong commitment to succeed are best practices for a successful integration with a materials management system. While integration may be complex, it is imperative to complete a materials management interface to achieve maximum value of an implant and supply management system. Integration to a materials management system leads to automation and increases productivity of the entire staff. Hospital staff can focus their valuable time on more important work, such as patient care, and have confidence that administrative tasks will be handled by iRISupply or other leading implant and supply management systems.
